The Ikoniscope20 fully-automated microscope

The FDA Cleared and CE marked Ikoniscope20 provides fully-automated slide handling, slide scanning, and real-time image capture and analysis, and eliminates many of the factors that cause human error in testing. It can scan and analyze up to 2,000,000 cells on a single slide, and as such, is designed for rare-cell detection based tests that cannot be performed manually due to the sheer number of cells that must be analyzed. The technology can also be used to automate tedious, labor intensive manual tests that can be undertaken in an efficient, reliable manner utilizing the Ikoniscope20.
Main features
The Ikoniscope Digital Microscope incorporates the necessary hardware and software to:
- - automatically load and scan cell and tissue slides (automation)
- - acquire fluorescence images of all the desired cells or tissue present on the slide and the diagnostic signals that they contain (digitization)
- - analyse and interpret the acquired images in a both qualitative and quantitative fashion; enumerating, classifying and presenting diagnostically relevant cells for review, without user intervention (analysis)
The platform incorporates the software needed to undertake these tasks in a fully hands-free fashion, including:
- - segmentation: determination of the boundaries of cell nuclei as imaged using a specific filter
- - signal detection: identifying and counting the number of chromosomes present in the interior of segmented nuclei, or identifying the presence of positive and negative antibody signals
- - auto focus: determination of the best-fit focus plane in a field image containing multiple cell nuclei at varying positions
- - auto exposure: determination of the best-fit exposure time in a field image containing a single signal channel expressed in multiple cell nuclei
- - auto matching: locating and orienting the deposition area on a FISH slide relative to an H&E (bright field) slide
Workflow
The Ikoniscope is designed for “walk-away” functionality, thus allowing the operator to focus their time on reviewing cells of interest instead of performing the otherwise labor-intensive step of sorting the cells that matters from the million of cells surrounding it.
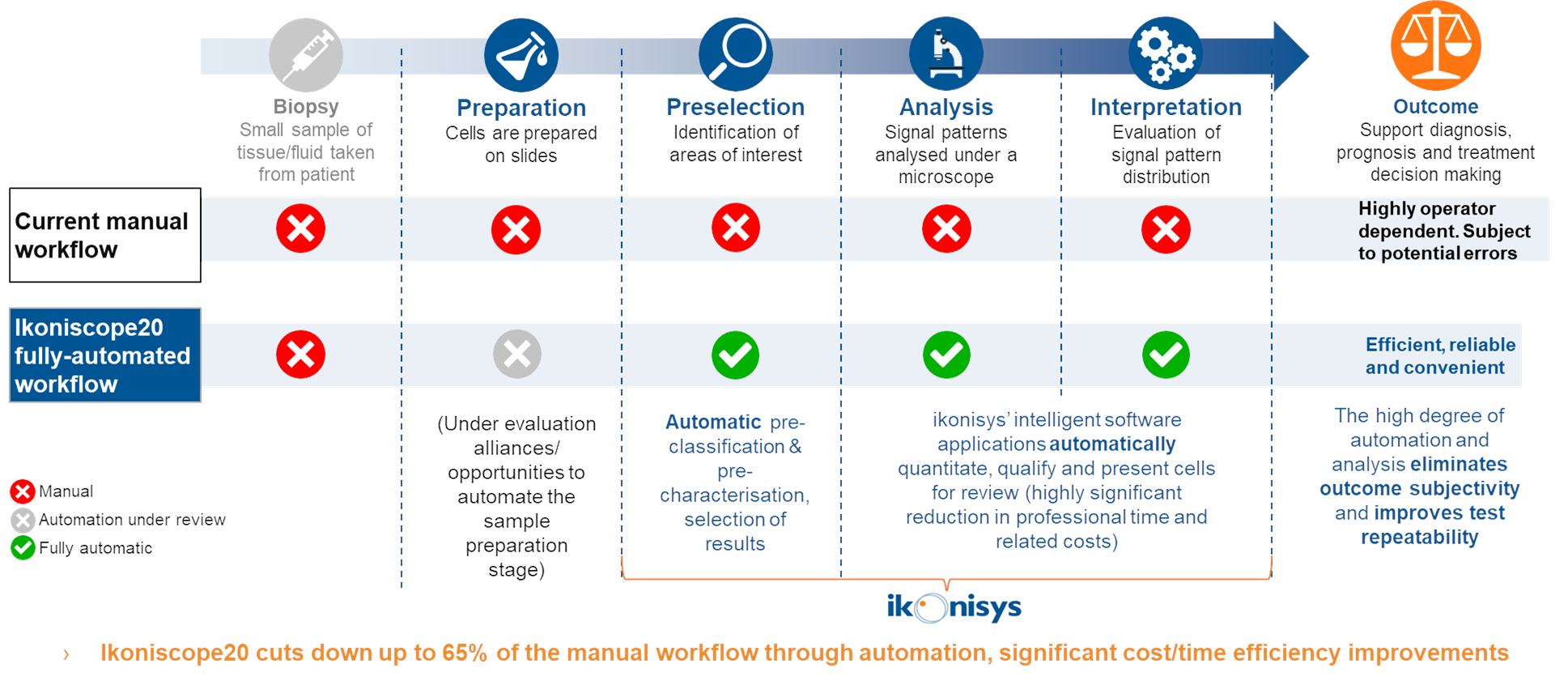
The Ikoniscope Digital Microscope drastically limits the number and duration of operator steps that have traditionally led to high labour costs and risks of error for MDx tests:
- - slides containing cell depositions or tissue samples and stained with the specific FISH Kit are loaded into multislide holding tray of the Ikoniscope20 in the scanning order desired. Patient data are entered and associated with each slide;
- - the Ikoniscope20 initially detects the cell deposition area, then scans the slide at low magnification. The results from a typical low magnification scan are depicted in the form of an overview image of the entire deposition area. Each cell within the deposition area is analysed to determine which are most likely to be abnormal;
- - image acquisition of the most likely abnormal cells is then undertaken at a higher magnification, in multiple focal planes. Each cell in the high magnification field image undergoes boundary segmentation and 3-dimensional FISH signal analysis;
- - on the basis of these results, the cell is classified into a specific category that ranges from normal to abnormal. All images and diagnostic information are stored for review;
- - a summary of the pertinent information for a slide scan is provided (including scan date and time and cell density). Also included is a data table with the number and percentage of cells that are classified into each category. The cells are ordered such that the most relevant ones are presented first;
- - images can be analysed in a gallery of nuclei or in the low- or high-magnification field. After analysing the slide scan images, the reviewer can export images of cells of interest and print a customizable report containing the outcome of the slide scan
A series of software products are available for the scanning and analysis of slides prepared with a variety of FISH probes.